Colored flames are a dazzling trick used by fire performers, in fireworks, and as a way to liven up campfires. The color that a burning substance emits is also important in the laboratory as the entire spectrum of light can be exhibited by flames depending on what temperature the flame is and what chemicals are being burned. We’ll discover the 9 different colors of flames and why they happen.
Why Are There Different Colors of Flames?

Different color flames are produced by burning specific substances.
©LYagovy/ via Getty Images
Flames are a process of something rapidly combining with oxygen and combusting. This is caused by an exothermic reaction which means that combining oxygen (O) and burning substances releases energy into its surroundings. Combustion creates light and heat through this release of energy.
The temperature and color of flames depend on what is combusting. The higher the heat at which a substance is burned is a part of determining what color the flame will be.
Within commonly seen fires, the hottest flames burn blue. As heat descends in temperature, flames appear white, yellow, orange, bright red, and then dark red. Burning specific things at the right temperature in the right situation creates flames with novel colors like pink, green, and black.
When an atom is heated, its electrons jump to a higher energy level than where they exist naturally. This is because the heat’s thermal energy becomes kinetic energy and causes the electrons to accelerate.
This acceleration is dramatic enough that it pushes electrons into a higher orbital than the one they’re usually in. When these excited electrons fall from their higher orbital back down to their normal orbital, they emit a photon.
These specific protons are often perceived as light by humans. Depending on the substance, it takes a certain amount of heat for light to be produced. This light varies based on what is burning.
1. Discovering White Flames

Burning Epsom salt, which contains magnesium, creates white flames.
©Martina Unbehauen/Shutterstock.com
When magnesium (Mg) burns, it creates white light. When substances containing beryllium (Be) are burned, they also create a white flame.
Magnesium sulfate (MgSO4), also known as Epsom salt, is the most commonly burned substance when white fire is desired outside of a laboratory. However, burning Epsom salt on a wood fire will not produce a white flame because the white flame is overpowered by the color of the combustion of the hydrocarbons present.
To achieve a white flame while burning Epsom salt, the initial fire needs to be fueled by something that is practically invisible when burned. These substances include ethanol, methanol, and isopropyl alcohol. This is how most white fire is created by fire performers.
2. Discovering Blue Flames

Blue flames are the hottest part of common flames.
©Wirestock/ via Getty Images
Purposefully manipulating flames to appear blue is done by burning cuprous chloride (CuCl) or more commonly butane (C4H10). The presence of arsenic (As) will produce a blue flame, and burning germanium (Ge) creates a pale blue flame.
Understanding blue flames is of particular interest when discussing firefighting. In smoldering wood, the surface of the burning object is between 842 to 1292 degrees Fahrenheit.
As the top of this range is exceeded, the radiating heat from the smoldering object creates a short blue flame that hugs the smoldering surface. This is an example of hydrocarbon burning in a unique environment, and for wood to get this hot, the surface must be smoldering charcoal. Blue flames outside of this situation usually don’t appear until an object is at least 2600 degrees Fahrenheit.
These blue flames happen because a critical radiation level has been exceeded which makes the immediate surroundings much hotter than the flammable object itself. This blue flame is what causes the spread of most house fires and wildfires as it is caused by irradiation. This means that as a fire burns, it heats surrounding objects to a point that they begin flaming independently. These independent flames start as a low-lying blue flame.
3. Discovering Yellow Flames

Most fires are predominantly yellow.
©Dogora Sun/Shutterstock.com
Yellow flames are the most common flames created by humans. It is the predominant color seen in campfires and candle flames. A typical yellow fire starts burning at 2100 degrees Fahrenheit.
The most common elements found in the molecules that create these yellow flames are iron (Fe) and sodium (Na). Sodium’s flame is so bright that it overwhelms the colors produced by other compounds that are burning. This makes most of the fire appear yellow.
4. Discovering Orange Flames

Fires are commonly associated with the color orange.
©iStock.com/Aleksei Nikolaev
Orange is one of the colors commonly associated with fire. That’s because most fires burn a hydrocarbon source like wood or charcoal. When carbon (C) burns, it appears bright orange.
When orange is artificially created by pyrotechnic experts, the chemical usually used is calcium chloride (CaCl2). Chemists create orange flames when they burn scandium (Sc).
5. Discovering Red Flames

Red fireworks are usually created with strontium chloride or strontium nitrate.
©maksime/ via Getty Images
In instances like fireworks where red is seen, the chemicals most likely causing this color are strontium chloride (SrCl2) or strontium nitrate (Sr(NO3)2). Cadmium (Cd) also burns brick red while mercury (Hg) creates a red flame.
Carmine is created when substances containing lithium (Li) are burned. Carmine is almost a crimson color, although it is slightly redder. Crimson is also obtained when radium (Ra) molecules are burned.
6. Discovering Green Flames
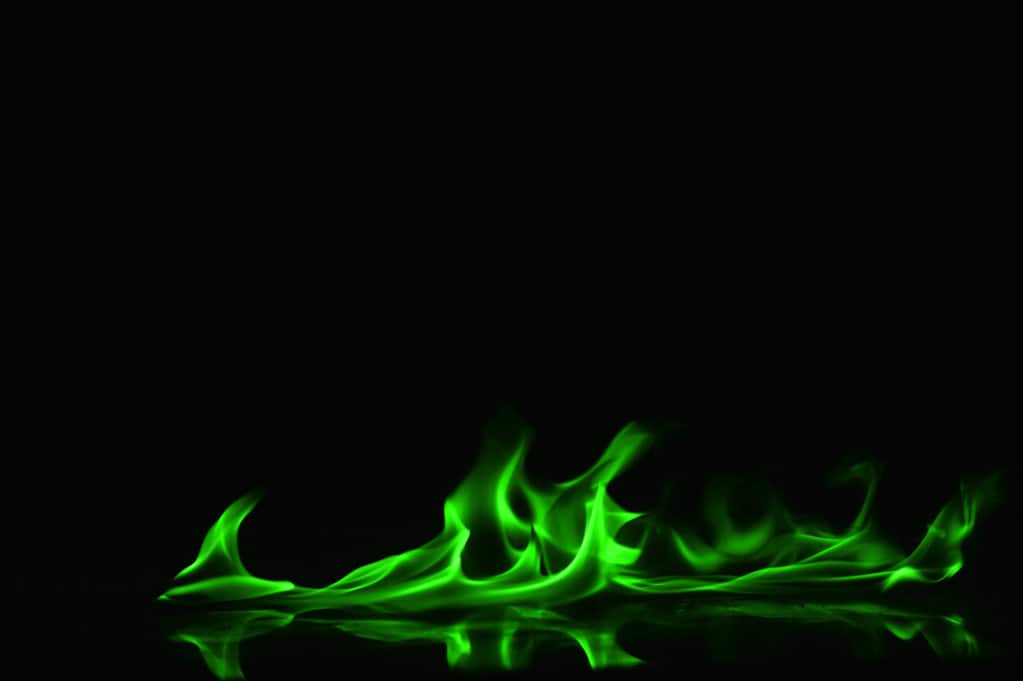
Borax and copper sulfate are common chemicals burned to produce green flames.
©piikcoro/ via Getty Images
Burning copper sulfate (CuSO4) or boric acid (B(OH)3) creates green flames. The presence of a substance containing barium (Ba) produces a pale green flame. Tungsten (W) molecules in laboratories burn green.
Sodium borate, commonly known as borax, creates a green flame despite sodium’s reputation to drown out other colors with yellow. This is because boron releases more energy than sodium as its electrons return to a less excited state.
7. Discovering Purple Flames and Why it Happens

Purple is not on the color spectrum so blue and red light mixes to create purple.
©mkos83/ via Getty Images
Purple flames are unique since purple is not a color naturally emitted. It is not on the color spectrum and it is a result of two colors mixing. Blue light and red light mixed create what we perceive as purple.
Purple is not a color that can be created on a wood-burning fire as the yellow in sodium will overpower any purple color created. Potassium chloride (KCl) or strontium nitrate (Sr(NO3)2) create a purple fire, and they need to be burned over a blue fire created by rubbing alcohol, ethanol, lighter fluid, or propane for the purple to be visible. Strontium nitrate is also used to create red flames in common yellow fires.
8. Discovering Pink Flames and Why it Happens

Pink is a color commonly used by pyrotechnic professionals even though it isn’t on the color spectrum.
©Chung Min/Shutterstock.com
Like purple fire, pink is not a color on the color spectrum. To achieve pink flames, red and white flames are combined so that their white wavelengths combine in a way that makes us perceive the color pink.
Burning lithium chloride (LiCl) creates a pink flame when burned by a substance containing magnesium. Mixtures of strontium nitrate and potassium nitrate (KNO3) burned with methanol are more commonly used by pyrotechnic professionals because these ingredients are easier to get.
9. Discovering Black Flames and Why it Happens

Light from low-pressure sodium lamps is required to create black flames.
©dzika_mrowka/ via Getty Images
Black is another color not naturally created by the wavelengths of light on the color spectrum. It has to be manipulated into existence by combining the wavelengths of multiple colors of light, or it appears in the absence of light. Black fire is one of the most complicated colors to obtain, so it isn’t used much in the ordinary world.
How to Create a Black Flame
A low-pressure sodium lamp is needed to create a black flame. These lamps contain sodium metal within the bulb that is vaporized. This produces a yellow light such as that commonly seen on roadside lighting or illuminating the underpasses of bridges.
A colorless flame created by burning methanol, ethanol, or rubbing alcohol is needed. Once the flame is created, salt water is dripped onto the flame. This creates a familiar yellow fire, but its intensity isn’t the same as that created by wood or candles. This means that the yellow will be overpowered by other introduced color wavelengths of light.
This alcohol and salt water fire needs to be created in an environment devoid of other light besides that created by a low-pressure sodium lamp. When sodium is added to the clear flame, the yellow from the dominating yellow of the environment created by the lamp absorbs the yellow created by the burning sodium of the flame. This makes the flame appear black in the absence of the flame’s light.
| Color of Flame | Element/Chemical Creating Flame | Temperatures that Create Flame (In Fahrenheit) | |
|---|---|---|---|
| 1. | Red Flame | SrCl2 or Sr(NO3)2 | 900 to 1800 Degrees |
| 2. | Orange Flame | C Molecules or CaCl2 | 1800 to 2100 Degrees |
| 3. | Yellow Flame | Fe or Na Molecules | 2100 to 2500 Degrees |
| 4. | White Flame | MgSO4 | 2500 to 2900 Degrees |
| 5. | Blue Flame | C4H10 or CuCl | 2600 to 3000 Degrees |
| 6. | Purple Flame | Combining Blue and Red Light | About 1800 Degrees |
| 7. | Green Flame | CuSO4 or B(OH)3 | 1800 to 2700 Degrees |
| 8. | Pink Flame | Combining Red and White Light | 2100 to 2400 Degrees |
| 9. | Black Flame | Manipulating Yellow Light | About 2300 Degrees |
The photo featured at the top of this post is © Deborah Kolb/Shutterstock.com
Thank you for reading! Have some feedback for us? Contact the AZ Animals editorial team.







