Helium — one of the noble gases — is odorless, tasteless, colorless, and has a unique profile when it comes to characteristics and behaviors. It’s also a very special gas among the noble gas group for a few different reasons. Namely, it has the largest ionization energy of any of the noble gases.
First, it’s important to understand what ionization energy is, and why helium needs it. Every element on the periodic table has a unique ionization energy, measured in kilojoules per mole.
Not only does it have the largest ionization; helium is the second-most abundant element in the universe behind hydrogen. Because it’s so abundant, nearly 25% of the universe is made up of helium! Helium atoms are so light they have the ability to escape Earth’s gravity — which is one of the reasons why it’s such a rare element on Earth.
Do you know why helium has the largest ionization energy? Keep reading to learn more about helium, ionization energy, and why helium has the largest ionization energy of any element!
The Secret Behind Helium’s High Ionization Energy
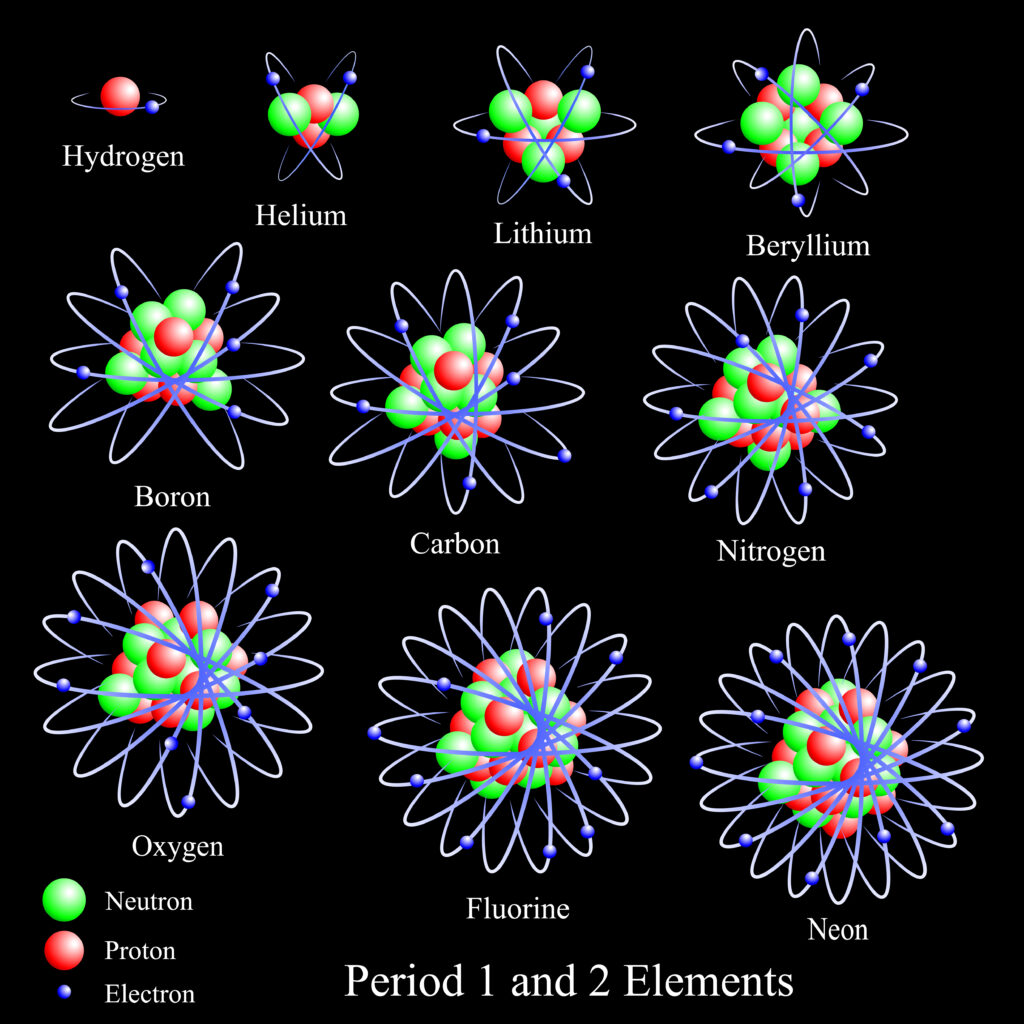
Helium only has two electrons orbiting its nucleus.
©Mike Price/Shutterstock.com
Helium has the largest ionization energy among all the elements because of its atomic structure. With just one electron shell outside of its nucleus, the energy it takes to ionize an electron away from the nucleus is the highest on the periodic table.
Why? First, it’s important to understand ionization energy.
The ionization energy of an atom or element depends on how strongly the nucleus holds onto the valence electron. Because helium has one electron shell with its only two electrons on it, it has a strong electrostatic attraction to the positively charged protons in the nucleus. As such, the electrons require a significant amount of energy to remove themselves from the atom completely. That’s why helium has the largest ionization energy among all the elements.
In general, ionization energy tends to increase as you move across a period (row) in the periodic table from left to right. This is because the number of protons in the nucleus increases, making the attraction between the nucleus and the electrons stronger, and therefore, it becomes more difficult to remove an electron.
Meet Helium: the Special Gas
You now know that helium is odorless, tasteless, and colorless — but did you know it’s also the second-lighting element on the periodic table? Not only that, helium is the only element that can’t be solidified by cooling at the normal atmospheric pressure all other elements can solidify at! Helium also has the lowest boiling point of any other known substance (−452.070 degrees Fahrenheit).
Unfortunately, helium is a non-renewable natural resource. This means once humans harvest all the naturally-occurring helium in the world, they’ll have to find an artificial way to recreate the element. Texas, Oklahoma, and Kansas have the most abundant helium around natural gas deposits.
Among helium’s other interesting properties, it has a high thermal conductivity and inertness, meaning helium can transfer heat well and does not readily react with other elements under normal conditions.
Compared to the other gases in the periodic table, helium sets itself apart with its atomic structure. It has two electrons in its outer shell while the rest of the noble gases have eight. Remember that valence electrons influence an atom’s ability to bond with another atom, thereby making something into a different compound or element. As such, helium’s valence electrons contribute to its high ionization energy.
Understanding Ionization Energy
By definition, ionization is the process of an atom becoming negatively or positively charged. How does this happen? It either gains or loses an electron, which has a negative charge. The resulting product of this change in electric charge is called an ion. So, the creation of an ion is called ionization.
Understanding how ions come to be helps to explain ionization energy. Ionization energy describes how much energy something requires to remove an electron from an atom or an ion to something else. Another way of thinking about ionization energy is how tightly an atom will hold onto its electrons. Atoms with high ionization energy want their electrons and work hard to keep them. Atoms with low ionization energy will readily give their electrons away without much resistance.
What Is the Order of the Ionization Energy?
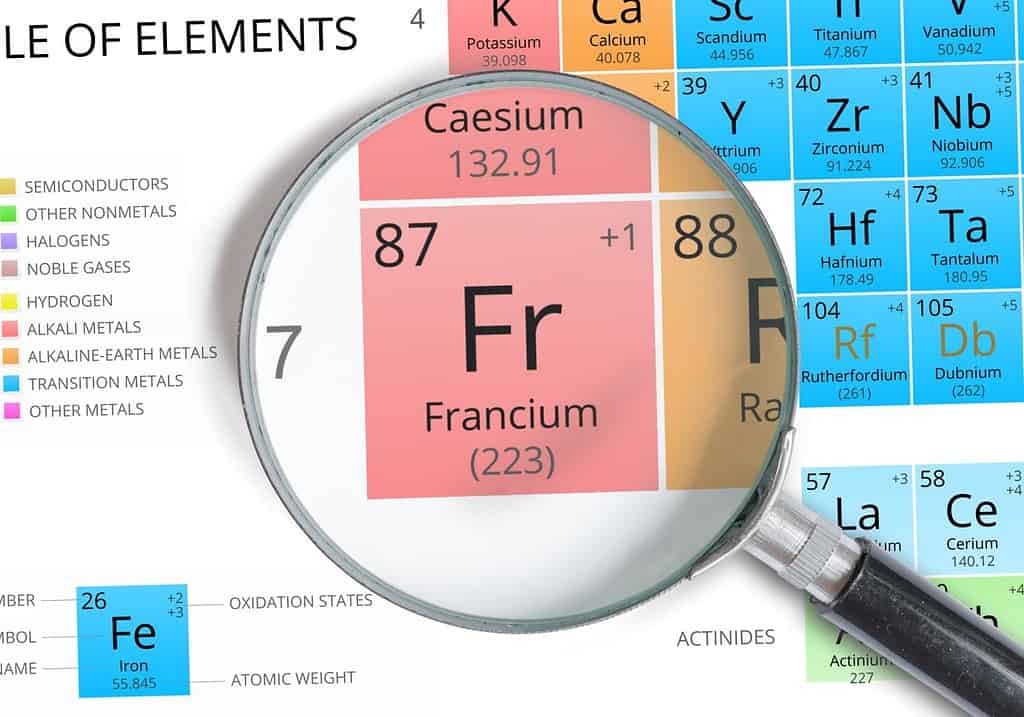
Francium has the lowest ionization energy because it has many shells and only one valence electron.
©vchal/Shutterstock.com
Understanding the order of ionization energy begins at the periodic table. Generally, the rule for ionization energy is: the highest energy is the top-most, right-most element on the table and the lowest is the bottom-most, left-most element based on the number of electron shells and valence electrons an element has. That makes helium the highest ionization energy and francium the lowest.
In addition to understanding how easily electrons can be removed from the atom, ionization energy gives scientists and researchers the ability to know how an element may react or behave. This is especially important in research that combines or wants to test the reactions of two or more elements.
Ionization energy is directly related to the number of electron shells. As a rule, elements with a higher number of shells have a lower ionization energy because the outer shell is so far away from the nucleus.
The Electron Configuration of Helium
First discovered in 1897, the electron has no known components or sub-particles. Simply put, it’s an elementary particle that acts as a building block to matter.
An atom is made up of several subatomic particles — one of which is an electron. Electrons have a charge of negative electricity, they are found in all atoms and enable the process of ionization. Electrons have the unique ability to balance an atom’s charge against the positive charge of protons in the atomic nucleus.
Electron configuration is a way of describing the arrangement of electrons in an atom. It involves using numbers and letters to represent how many electrons are in each level or “shell” of the atom. The electron configuration helps to understand how electrons behave and why some elements interact with others while others do not. Scientists use this information to study and comprehend the characteristics of various atoms.
Real-Life Applications of Helium’s Special Properties

Because helium is unreactive (unlike hydrogen) it makes sense to use it in items like balloons.
©iStock.com/anyaberkut
Helium is most often used in balloons. Why? Well, helium’s special properties allow balloons to:
- Be lighter than air.
- Not combust (like hydrogen would).
- Be used safely (it’s non-toxic).
In addition to blowing up balloons, helium is used in airships and blimps and in cooling systems. On airships, helium functions similarly to how it’s used in a balloon. Because helium is non-combustible and lighter than air, it has a great lifting capacity and doesn’t burn — which could result in fatalities to anyone on board. Helium’s thermal conductivity makes it a great cooling agent because it doesn’t solidify under extreme temperatures. Machines and technology that use helium cooling agents include satellite instruments, superconductive magnet coils, and more.
Finally, helium’s small size and inert nature make it an excellent tracer gas for detecting leaks in various systems, such as pipelines, containers, and air conditioning units. Helium-oxygen mixtures, known as heliox, are used for deep-sea diving to reduce the risk of nitrogen narcosis, a condition caused by high nitrogen levels in the bloodstream.
Helium Has the Highest Ionization Energy on the Periodic Table
Helium has the largest ionization energy because of its atomic structure. With its outer electrons holding so tightly to the center, it takes a massive amount of energy to completely remove it from the atom it orbits.
Now that you have a good understanding of helium’s unique properties and why it has the largest ionization energy of all the elements, you can learn more about the world.
Thank you for reading! Have some feedback for us? Contact the AZ Animals editorial team.








